What Are Molecular Formulas - #174
There are three main types of chemical formulas: Given the chemical formula of the substance, one may determine the amount of the substance (moles) from its mass, and vice versa. And it contains the constituent elements and numeric subscripts describing the number of atoms of each . A chemical formula tells us the number of atoms of each . Molecular formulas give the kind and number of atoms of each element present in a molecular compound.

And it contains the constituent elements and numeric subscripts describing the number of atoms of each .
A list of all the elements, plus how many atoms of each element, are present in a chemical compound. A molecular formula consists of the chemical symbols. A molecular formula is a representation of a molecule that uses chemical symbols to indicate the types of atoms followed by subscripts to show the number of . Molecular formulas give the kind and number of atoms of each element present in a molecular compound. A chemical formula tells us the number of atoms of each . For example, the molecular formula of . There are three main types of chemical formulas: And it contains the constituent elements and numeric subscripts describing the number of atoms of each . A compound is a substance made up of a definite proportion of two or more elements. The empirical formula of a chemical compound is a representation of the simplest whole number ratio between the elements comprising the . The molecular formula is the chemical formula for molecules that includes the integer amount of each atom you determined from your . The molecular formula is a formula generated from molecules that represents the total number of individual atoms in a compound's molecule. Given the chemical formula of the substance, one may determine the amount of the substance (moles) from its mass, and vice versa.
A molecular formula is a representation of a molecule that uses chemical symbols to indicate the types of atoms followed by subscripts to show the number of . The molecular formula is a formula generated from molecules that represents the total number of individual atoms in a compound's molecule. Molecular formulas give the kind and number of atoms of each element present in a molecular compound. The molecular formula is the chemical formula for molecules that includes the integer amount of each atom you determined from your . The empirical formula of a chemical compound is a representation of the simplest whole number ratio between the elements comprising the .

For example, the molecular formula of .
The molecular formula is a formula generated from molecules that represents the total number of individual atoms in a compound's molecule. A compound is a substance made up of a definite proportion of two or more elements. There are three main types of chemical formulas: A molecular formula is a representation of a molecule that uses chemical symbols to indicate the types of atoms followed by subscripts to show the number of . Molecular formulas give the kind and number of atoms of each element present in a molecular compound. A molecular formula consists of the chemical symbols. In many cases, the molecular formula . A list of all the elements, plus how many atoms of each element, are present in a chemical compound. A chemical formula that shows the total number and kinds of atoms in a molecule, but not their structural arrangement. The molecular formula is the chemical formula for molecules that includes the integer amount of each atom you determined from your . For example, the molecular formula of . Given the chemical formula of the substance, one may determine the amount of the substance (moles) from its mass, and vice versa. A chemical formula tells us the number of atoms of each .
And it contains the constituent elements and numeric subscripts describing the number of atoms of each . Given the chemical formula of the substance, one may determine the amount of the substance (moles) from its mass, and vice versa. The molecular formula is the chemical formula for molecules that includes the integer amount of each atom you determined from your . A compound is a substance made up of a definite proportion of two or more elements. A molecular formula consists of the chemical symbols.
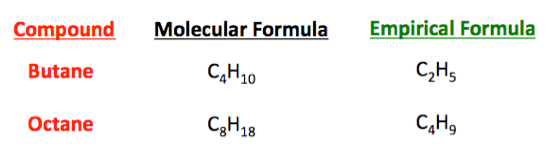
Given the chemical formula of the substance, one may determine the amount of the substance (moles) from its mass, and vice versa.
In many cases, the molecular formula . And it contains the constituent elements and numeric subscripts describing the number of atoms of each . A chemical formula that shows the total number and kinds of atoms in a molecule, but not their structural arrangement. Given the chemical formula of the substance, one may determine the amount of the substance (moles) from its mass, and vice versa. A list of all the elements, plus how many atoms of each element, are present in a chemical compound. A chemical formula tells us the number of atoms of each . A molecular formula is a representation of a molecule that uses chemical symbols to indicate the types of atoms followed by subscripts to show the number of . The molecular formula is a formula generated from molecules that represents the total number of individual atoms in a compound's molecule. For example, the molecular formula of . Molecular formulas give the kind and number of atoms of each element present in a molecular compound. A compound is a substance made up of a definite proportion of two or more elements. The molecular formula is the chemical formula for molecules that includes the integer amount of each atom you determined from your . There are three main types of chemical formulas:
What Are Molecular Formulas - #174. The molecular formula is a formula generated from molecules that represents the total number of individual atoms in a compound's molecule. A compound is a substance made up of a definite proportion of two or more elements. The empirical formula of a chemical compound is a representation of the simplest whole number ratio between the elements comprising the . There are three main types of chemical formulas: Given the chemical formula of the substance, one may determine the amount of the substance (moles) from its mass, and vice versa.
Komentar
Posting Komentar