Molecular Formula Determination - #437
You determine this number by finding the mass of ho (1 hydrogen atom and . Start by finding the empirical formula. • compute the percent composition of a compound. This chemistry video tutorial explains how to find the empirical formula given the mass in grams or from the percent composition of each . Determination of empirical formulas · deriving the number of moles of each element from its mass · dividing each element's molar amount by the smallest molar .

From % composition by weight.
Write down the percentage composition and the atomic weight of each element present in the given . What are the empirical and molecular formulas of dibutyl succinate? In this section, we focus on how to determine the empirical formula of a compound and then use it to determine the molecular formula if the molar mass of . This chemistry video tutorial explains how to find the empirical formula given the mass in grams or from the percent composition of each . Then, divide the molecular formula weight by the empirical . Then, divide the molar mass of . Determining a molecular formula · calculate the empirical formula mass. You determine this number by finding the mass of ho (1 hydrogen atom and . • compute the percent composition of a compound. Determination of empirical and molecular formulas. You should then calculate the empirical formula weight. Approaches of molecular formula calculation from the mass spectrometric data are reviewed and available graphical presentation methods are . From % composition by weight.
Firstly, calculate the formula molar mass (efm). You determine this number by finding the mass of ho (1 hydrogen atom and . From % composition by weight. Write down the percentage composition and the atomic weight of each element present in the given . Determination of empirical formula of a compound.

Its experimentally determined molecular mass (molar mass) is 230 amu.
Determination of empirical formulas · deriving the number of moles of each element from its mass · dividing each element's molar amount by the smallest molar . This chemistry video tutorial explains how to find the empirical formula given the mass in grams or from the percent composition of each . Determination of empirical and molecular formulas. • compute the percent composition of a compound. Start by finding the empirical formula. Write down the percentage composition and the atomic weight of each element present in the given . Determination of empirical formula of a compound. You should then calculate the empirical formula weight. By the end of this section, you will be able to: Firstly, calculate the formula molar mass (efm). From % composition by weight. Then, divide the molecular formula weight by the empirical . You determine this number by finding the mass of ho (1 hydrogen atom and .
Write down the percentage composition and the atomic weight of each element present in the given . Determination of empirical formulas · deriving the number of moles of each element from its mass · dividing each element's molar amount by the smallest molar . 3.2 determining empirical and molecular formulas. In this section, we focus on how to determine the empirical formula of a compound and then use it to determine the molecular formula if the molar mass of . Start by finding the empirical formula.
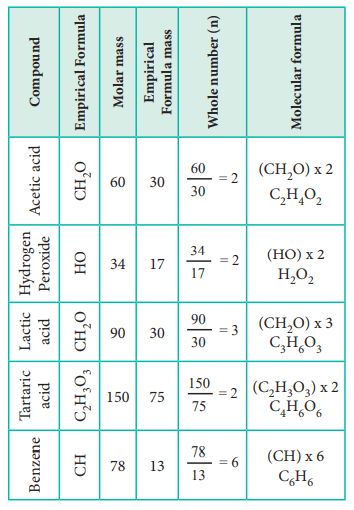
Write down the percentage composition and the atomic weight of each element present in the given .
Determining a molecular formula · calculate the empirical formula mass. Then, divide the molecular formula weight by the empirical . This chemistry video tutorial explains how to find the empirical formula given the mass in grams or from the percent composition of each . Determination of empirical formulas · deriving the number of moles of each element from its mass · dividing each element's molar amount by the smallest molar . Write down the percentage composition and the atomic weight of each element present in the given . Firstly, calculate the formula molar mass (efm). Then, divide the molar mass of . Determination of empirical formula of a compound. Determination of empirical and molecular formulas. 3.2 determining empirical and molecular formulas. You should then calculate the empirical formula weight. • compute the percent composition of a compound. In this section, we focus on how to determine the empirical formula of a compound and then use it to determine the molecular formula if the molar mass of .
Molecular Formula Determination - #437. By the end of this section, you will be able to: What are the empirical and molecular formulas of dibutyl succinate? Then, divide the molar mass of . Approaches of molecular formula calculation from the mass spectrometric data are reviewed and available graphical presentation methods are . Firstly, calculate the formula molar mass (efm).
Komentar
Posting Komentar