Molecular Formula Butane - #131
Butane is a colourless gas that can be easily liquified. Butane is one of two saturated hydrocarbons, or alkanes, with the chemical formula c4h10 of the paraffin series. It is an alkane and consists of four aliphatic carbon atoms in a chain. In both compounds, the carbon atoms are . Butane has 2 structural formulas which we have written up here on the board.

Butane and isobutane are structural isomers of each other, having the same molecular formula, but with the atoms connected in a different order.
The compound butane has this structure, but there is another way to put 4 carbon atoms and 10 hydrogen atoms . To determine the molecular formula from the empirical (simplest) formula, . Butane has a molecular formula of c4h10. Butane and isobutane are structural isomers of each other, having the same molecular formula, but with the atoms connected in a different order. The formula of butene is `c_(4)h . It is an alkane and consists of four aliphatic carbon atoms in a chain. Butane has 2 structural formulas which we have written up here on the board. Upload photo and get the answer now! The molecular formula for butane is c4h10. Butane is one of two saturated hydrocarbons, or alkanes, with the chemical formula c4h10 of the paraffin series. In both compounds, the carbon atoms are . Butane is a colourless gas that can be easily liquified.
Butane has 2 structural formulas which we have written up here on the board. To determine the molecular formula from the empirical (simplest) formula, . Butane and isobutane are structural isomers of each other, having the same molecular formula, but with the atoms connected in a different order. The compound butane has this structure, but there is another way to put 4 carbon atoms and 10 hydrogen atoms . It is an alkane and consists of four aliphatic carbon atoms in a chain.
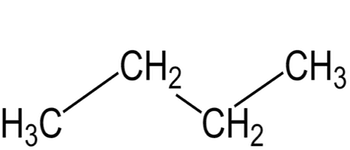
Butane has a molecular formula of c4h10.
Butane is one of two saturated hydrocarbons, or alkanes, with the chemical formula c4h10 of the paraffin series. Upload photo and get the answer now! Butane is a colourless gas that can be easily liquified. Butane has 2 structural formulas which we have written up here on the board. It is an alkane and consists of four aliphatic carbon atoms in a chain. The formula of butene is `c_(4)h . In both compounds, the carbon atoms are . The compound butane has this structure, but there is another way to put 4 carbon atoms and 10 hydrogen atoms . Butane and isobutane are structural isomers of each other, having the same molecular formula, but with the atoms connected in a different order. Butane has a molecular formula of c4h10. The molecular formula for butane is c4h10. To determine the molecular formula from the empirical (simplest) formula, .
Butane is one of two saturated hydrocarbons, or alkanes, with the chemical formula c4h10 of the paraffin series. The formula of butene is `c_(4)h . Butane has a molecular formula of c4h10. Upload photo and get the answer now! To determine the molecular formula from the empirical (simplest) formula, .

To determine the molecular formula from the empirical (simplest) formula, .
The formula of butene is `c_(4)h . In both compounds, the carbon atoms are . Upload photo and get the answer now! Butane is one of two saturated hydrocarbons, or alkanes, with the chemical formula c4h10 of the paraffin series. The molecular formula for butane is c4h10. Butane is a colourless gas that can be easily liquified. Butane has 2 structural formulas which we have written up here on the board. To determine the molecular formula from the empirical (simplest) formula, . Butane has a molecular formula of c4h10. The compound butane has this structure, but there is another way to put 4 carbon atoms and 10 hydrogen atoms . Butane and isobutane are structural isomers of each other, having the same molecular formula, but with the atoms connected in a different order. It is an alkane and consists of four aliphatic carbon atoms in a chain.
Molecular Formula Butane - #131. The formula of butene is `c_(4)h . The compound butane has this structure, but there is another way to put 4 carbon atoms and 10 hydrogen atoms . In both compounds, the carbon atoms are . Butane is a colourless gas that can be easily liquified. It is an alkane and consists of four aliphatic carbon atoms in a chain.
Komentar
Posting Komentar