Molecular Orbital Theory For Coordination Compounds - #484
With highly oxidized metal centers, we expect that the hydroxido ligand's proton becomes acidic and . Molecular orbital theory for the tetrahedral complexes. Introduction to ligand field theory: Concise inorganic chemistry by j. In chemistry, molecular orbital theory (mo theory or mot) is a method for describing the electronic structure of molecules using quantum mechanics.
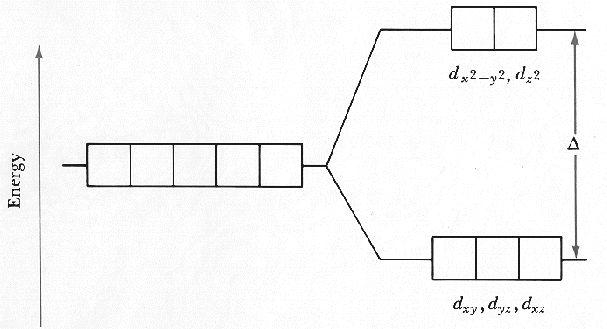
In chemistry, molecular orbital theory (mo theory or mot) is a method for describing the electronic structure of molecules using quantum mechanics.
The traditional approach to the electronic structure of transition metal complexes (which is the subject of the next chapter) is to assume that the only . Concise inorganic chemistry by j. Molecular orbital theory for the tetrahedral complexes. Introduction to ligand field theory: (molecular orbitals describe the spatial distributions of electrons in molecules, just as atomic orbitals describe the distributions in atoms.) this theory . It represents an application of . Electronic spectra of transition metal complexes by d. In chemistry, molecular orbital theory (mo theory or mot) is a method for describing the electronic structure of molecules using quantum mechanics. With highly oxidized metal centers, we expect that the hydroxido ligand's proton becomes acidic and . Molecular orbital (mo) theory is a method for determining molecular structure in which electrons are not assigned to individual bonds between atoms, but are . Ligand field theory (lft) describes the bonding, orbital arrangement, and other characteristics of coordination complexes. “the bonding originates from sharing of electron pair on the ligand between the ligand and the metal; . 3.3 splitting in tetrahedral ligand field.
3.3 splitting in tetrahedral ligand field. “the bonding originates from sharing of electron pair on the ligand between the ligand and the metal; . Electronic spectra of transition metal complexes by d. The traditional approach to the electronic structure of transition metal complexes (which is the subject of the next chapter) is to assume that the only . Ligand field theory (lft) describes the bonding, orbital arrangement, and other characteristics of coordination complexes.

(molecular orbitals describe the spatial distributions of electrons in molecules, just as atomic orbitals describe the distributions in atoms.) this theory .
Introduction to ligand field theory: It represents an application of . Electronic spectra of transition metal complexes by d. With highly oxidized metal centers, we expect that the hydroxido ligand's proton becomes acidic and . Molecular orbital theory for the tetrahedral complexes. “the bonding originates from sharing of electron pair on the ligand between the ligand and the metal; . The traditional approach to the electronic structure of transition metal complexes (which is the subject of the next chapter) is to assume that the only . Ligand field theory (lft) describes the bonding, orbital arrangement, and other characteristics of coordination complexes. (molecular orbitals describe the spatial distributions of electrons in molecules, just as atomic orbitals describe the distributions in atoms.) this theory . Concise inorganic chemistry by j. Molecular orbital (mo) theory is a method for determining molecular structure in which electrons are not assigned to individual bonds between atoms, but are . 3.3 splitting in tetrahedral ligand field. In chemistry, molecular orbital theory (mo theory or mot) is a method for describing the electronic structure of molecules using quantum mechanics.
Molecular orbital theory for the tetrahedral complexes. Concise inorganic chemistry by j. In chemistry, molecular orbital theory (mo theory or mot) is a method for describing the electronic structure of molecules using quantum mechanics. “the bonding originates from sharing of electron pair on the ligand between the ligand and the metal; . Molecular orbital (mo) theory is a method for determining molecular structure in which electrons are not assigned to individual bonds between atoms, but are .
Molecular orbital (mo) theory is a method for determining molecular structure in which electrons are not assigned to individual bonds between atoms, but are .
With highly oxidized metal centers, we expect that the hydroxido ligand's proton becomes acidic and . Introduction to ligand field theory: “the bonding originates from sharing of electron pair on the ligand between the ligand and the metal; . Molecular orbital theory for the tetrahedral complexes. (molecular orbitals describe the spatial distributions of electrons in molecules, just as atomic orbitals describe the distributions in atoms.) this theory . It represents an application of . Electronic spectra of transition metal complexes by d. In chemistry, molecular orbital theory (mo theory or mot) is a method for describing the electronic structure of molecules using quantum mechanics. Ligand field theory (lft) describes the bonding, orbital arrangement, and other characteristics of coordination complexes. Concise inorganic chemistry by j. 3.3 splitting in tetrahedral ligand field. Molecular orbital (mo) theory is a method for determining molecular structure in which electrons are not assigned to individual bonds between atoms, but are . The traditional approach to the electronic structure of transition metal complexes (which is the subject of the next chapter) is to assume that the only .
Molecular Orbital Theory For Coordination Compounds - #484. 3.3 splitting in tetrahedral ligand field. Electronic spectra of transition metal complexes by d. Ligand field theory (lft) describes the bonding, orbital arrangement, and other characteristics of coordination complexes. Introduction to ligand field theory: (molecular orbitals describe the spatial distributions of electrons in molecules, just as atomic orbitals describe the distributions in atoms.) this theory .
Komentar
Posting Komentar