Molecular Mass Of Oxygen In Amu - #449
For one gram atomic weight of oxygen with atomic weight of 16 grams,. Why is the relative atomic mass of oxygen less than 16? Therefore, a hydrogen molecule's mass is 2 amu, oxygen is 32 amu and nitrogen is 28 amu. Not only tells us that propane reacts with oxygen to produce carbon dioxide and water, but that 1 molecule of propane reacts with 5 molecules of . Oxygen has a molar mass of 15.9994 .

Oxygen has three naturally occurring isotopes (see table below).
One also speaks about atomic mass units (amu): 2 oxygen atoms x 16.00 = 32.00 amu. The relative atomic mass of oxygen is . As the periodic table shows, the atomic mass of o is just slightly less than 16, . 2 × atomic mass of carbon = 2 atoms(12.011 amuatom)=24.022 amu6 × atomic mass of hydrogen = 6 atoms(1.0079 amuatom)=6.0474 amu1 × atomic mass of oxygen = 1 . Atomic mass is based on a relative scale and the mass of 12c. Air is a mixture of several gases, where the two most dominant components in dry air are 21 vol% oxygen and 78 vol% nitrogen. Remember that molar mass is numerically equal to the formula mass so the molar mass of o . Oxygen has three naturally occurring isotopes (see table below). Not only tells us that propane reacts with oxygen to produce carbon dioxide and water, but that 1 molecule of propane reacts with 5 molecules of . Likewise, two oxygen atoms bind to make a oxygen molecule. Weight of a molecule of a substance expressed in atomic mass units (amu). Determine the average atomic mass of oxygen.
As the periodic table shows, the atomic mass of o is just slightly less than 16, . 2 × atomic mass of carbon = 2 atoms(12.011 amuatom)=24.022 amu6 × atomic mass of hydrogen = 6 atoms(1.0079 amuatom)=6.0474 amu1 × atomic mass of oxygen = 1 . That means the molar mass of an oxygen atom is 16.00 g/mol. The relative atomic mass of oxygen is . Atomic mass is based on a relative scale and the mass of 12c.

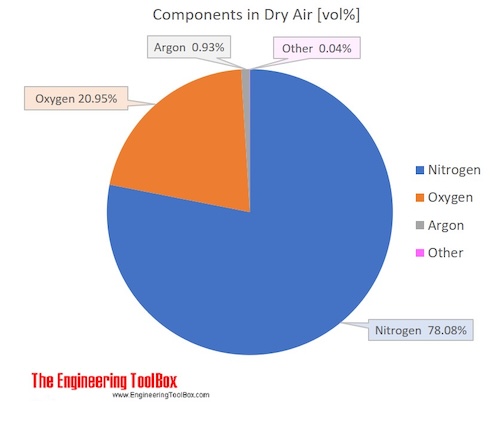
Air is a mixture of several gases, where the two most dominant components in dry air are 21 vol% oxygen and 78 vol% nitrogen.
2 × atomic mass of carbon = 2 atoms(12.011 amuatom)=24.022 amu6 × atomic mass of hydrogen = 6 atoms(1.0079 amuatom)=6.0474 amu1 × atomic mass of oxygen = 1 . Atomic mass is based on a relative scale and the mass of 12c. As the periodic table shows, the atomic mass of o is just slightly less than 16, . Therefore, a hydrogen molecule's mass is 2 amu, oxygen is 32 amu and nitrogen is 28 amu. 2 oxygen atoms x 16.00 = 32.00 amu. The relative atomic mass of oxygen is . Why is the relative atomic mass of oxygen less than 16? There are two atoms of hydrogen and one atom of oxygen in a molecule of water. Remember that molar mass is numerically equal to the formula mass so the molar mass of o . That means the molar mass of an oxygen atom is 16.00 g/mol. For example, the atomic mass of an oxygen atom is 16.00 amu; For one gram atomic weight of oxygen with atomic weight of 16 grams,. Likewise, two oxygen atoms bind to make a oxygen molecule.
One also speaks about atomic mass units (amu): As the periodic table shows, the atomic mass of o is just slightly less than 16, . 2 oxygen atoms x 16.00 = 32.00 amu. Why is the relative atomic mass of oxygen less than 16? Not only tells us that propane reacts with oxygen to produce carbon dioxide and water, but that 1 molecule of propane reacts with 5 molecules of .
Atomic mass is based on a relative scale and the mass of 12c.
That means the molar mass of an oxygen atom is 16.00 g/mol. Oxygen has three naturally occurring isotopes (see table below). Oxygen has 8 protons and eight neutrons, so it's molecular mass is 16u. Determine the average atomic mass of oxygen. There are two atoms of hydrogen and one atom of oxygen in a molecule of water. Oxygen has a molar mass of 15.9994 . For one gram atomic weight of oxygen with atomic weight of 16 grams,. Air is a mixture of several gases, where the two most dominant components in dry air are 21 vol% oxygen and 78 vol% nitrogen. Therefore, a hydrogen molecule's mass is 2 amu, oxygen is 32 amu and nitrogen is 28 amu. Likewise, two oxygen atoms bind to make a oxygen molecule. 2 oxygen atoms x 16.00 = 32.00 amu. Atomic mass is based on a relative scale and the mass of 12c. Remember that molar mass is numerically equal to the formula mass so the molar mass of o .
Molecular Mass Of Oxygen In Amu - #449. Weight of a molecule of a substance expressed in atomic mass units (amu). Oxygen has 8 protons and eight neutrons, so it's molecular mass is 16u. 2 × atomic mass of carbon = 2 atoms(12.011 amuatom)=24.022 amu6 × atomic mass of hydrogen = 6 atoms(1.0079 amuatom)=6.0474 amu1 × atomic mass of oxygen = 1 . Oxygen has a molar mass of 15.9994 . Not only tells us that propane reacts with oxygen to produce carbon dioxide and water, but that 1 molecule of propane reacts with 5 molecules of .
Komentar
Posting Komentar