Molecular Mass Meaning - #248
This leads to two important facts. The molecular mass (m) is the mass of a given molecule: Molar mass is the mass equivalent of avogadro's number of atoms of an element, or avogadro's number of molecules of in a chemical compound. It is usually measured in daltons (da or u).different molecules of the same compound may have different molecular masses … The related quantity relative molecular mass, as defined by iupac, is the ratio of the mass of a molecule to the unified atomic mass unit (also known as the dalton) and is unitless.
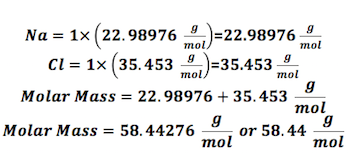
Mass of a mole of a substance.
In chemistry, it is used to determine … 26/09/2021 · the molecular mass of an element is defined as the sum of all the masses of the elements present inside the molecule. It helps to determine … Molar mass is the mass equivalent of avogadro's number of atoms of an element, or avogadro's number of molecules of in a chemical compound. The molecular mass and relative molecular mass are distinct from but rel… This leads to two important facts. Molecular mass is a measure of the sum of the atomic masses of the atoms in a molecule. It is usually measured in daltons (da or u).different molecules of the same compound may have different molecular masses … It’s also referred to as molecular weight. The molecular mass (m) is the mass of a given molecule: Mass of a mole of a substance. Mo·lec·u·lar weight (mol wt, mw), the sum of the atomic weights of all the atoms constituting a molecule; The molecular mass (m) is the mass of a given molecule:
The molecular mass of water, in which there are … It’s also referred to as molecular weight. It helps to determine … Mass of a mole of a substance. The related quantity relative molecular mass, as defined by iupac, is the ratio of the mass of a molecule to the unified atomic mass unit (also known as the dalton) and is unitless.

The related quantity relative molecular mass, as defined by iupac, is the ratio of the mass of a molecule to the unified atomic mass unit (also known as the dalton) and is unitless.
The molecular mass (m) is the mass of a given molecule: It’s also referred to as molecular weight. It helps to determine … The molecular mass of water, in which there are … Molar mass is the mass equivalent of avogadro's number of atoms of an element, or avogadro's number of molecules of in a chemical compound. Different molecules of the same compound may have different molecular masses because they contain different isotopes of an element. noun the mass of a molecule that is equal to the sum of the masses of all the atoms contained in the molecule. It is measured in daltons (da or u). This leads to two important facts. What is molecular mass used for? The molecular mass and relative molecular mass are distinct from but rel… Mo·lec·u·lar weight (mol wt, mw), the sum of the atomic weights of all the atoms constituting a molecule; In chemistry, it is used to determine …
26/09/2021 · the molecular mass of an element is defined as the sum of all the masses of the elements present inside the molecule. Different molecules of the same compound may have different molecular masses because they contain different isotopes of an element. It is usually measured in daltons (da or u).different molecules of the same compound may have different molecular masses … The molecular mass (m) is the mass of a given molecule: It’s also referred to as molecular weight.

What is molecular mass used for?
noun the mass of a molecule that is equal to the sum of the masses of all the atoms contained in the molecule. Mo·lec·u·lar weight (mol wt, mw), the sum of the atomic weights of all the atoms constituting a molecule; Molar mass is the mass equivalent of avogadro's number of atoms of an element, or avogadro's number of molecules of in a chemical compound. The mass of a molecule relative to the mass of a standard atom, now 12c (taken … The molecular mass of water, in which there are … Molecular mass is a measure of the sum of the atomic masses of the atoms in a molecule. In chemistry, it is used to determine … 21/09/2017 · molar mass = mass/mole = g/mol. Different molecules of the same compound may have different molecular masses because they contain different isotopes of an element. It is measured in daltons (da or u). What is molecular mass used for? The molecular mass (m) is the mass of a given molecule: It is usually measured in daltons (da or u).different molecules of the same compound may have different molecular masses …
Molecular Mass Meaning - #248. In chemistry, it is used to determine … 21/09/2017 · molar mass = mass/mole = g/mol. It is usually measured in daltons (da or u).different molecules of the same compound may have different molecular masses … Molar mass is the mass equivalent of avogadro's number of atoms of an element, or avogadro's number of molecules of in a chemical compound. The molecular mass and relative molecular mass are distinct from but rel…
Komentar
Posting Komentar