Molecular Mass Hydrogen - #302
The structure of molecular hydrogen is h−h . The molecular weight of a hydrogen molecule is m=2.016×10−3 kg/mol. Hydrogen (h), 1.00794 g/mol ; Rounded to three decimal places, the atomic weight of hydrogen is 1.008 amu and that of oxygen is 15.999 amu. The molar mass of a substance is the mass in grams of 1 mole of the substance.
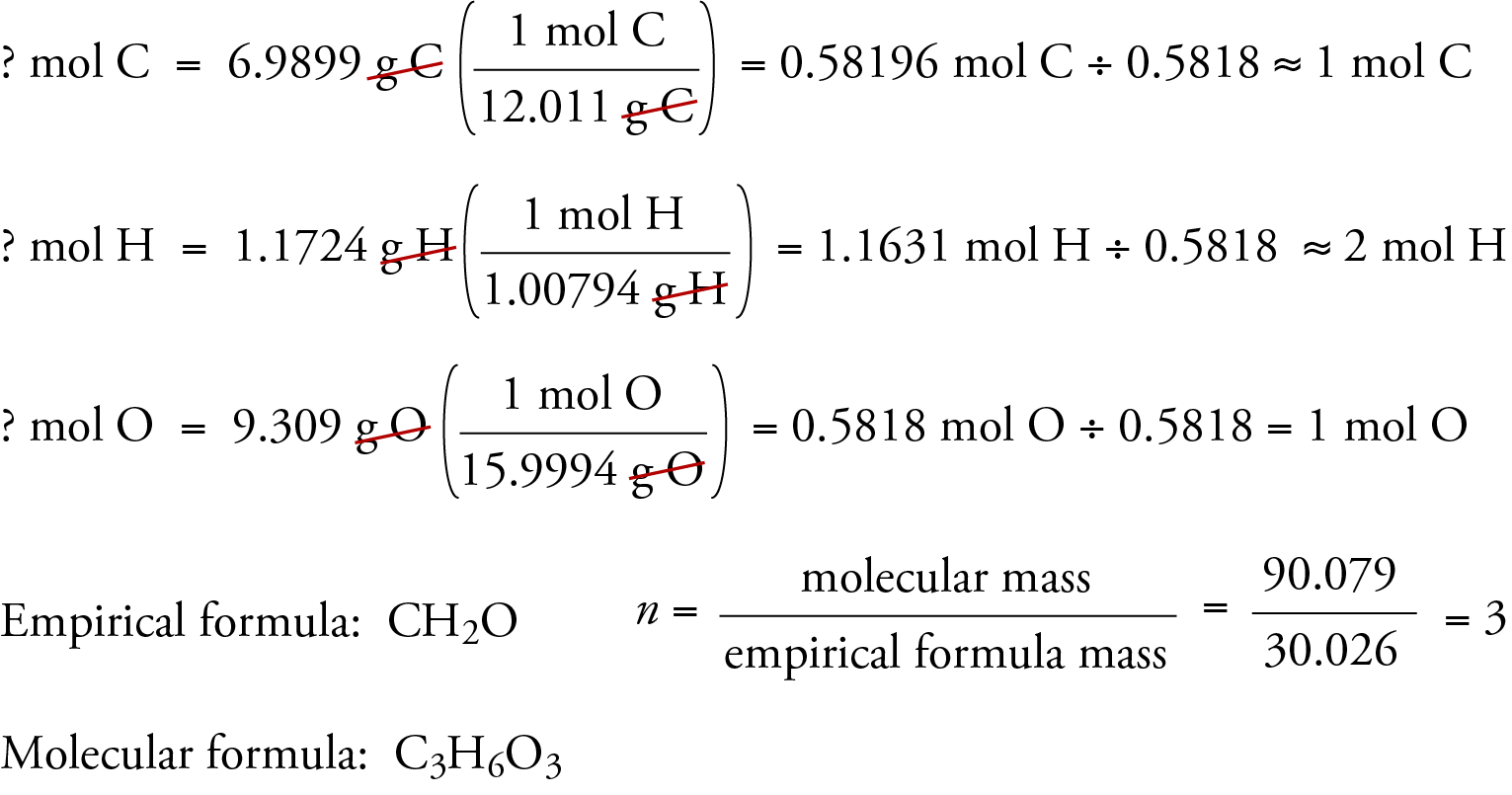
Carbon (c), 12.0107 g/mol ;
So, one more electron is needed to complete its octet. Iron (fe), 55.845 g/mol ; The molecular weight of a hydrogen molecule is m=2.016×10−3 kg/mol. 1, h · hydrogen, 1.00797 ; Hydrogen molar mass is 1.00794 g/mol · the molar mass of the hydrogen molecule is 2.016 g/mol. The molecular weight of water is thus (2×1.008)+(1 . The sum of the atomic masses of all atoms in a molecule, based on a scale in which the atomic masses of hydrogen, carbon, nitrogen, and oxygen are 1, 12, . Using the periodic table of the elements to find atomic weights, we find that hydrogen has an atomic weight of 1, and oxygen's is 16. Definition and molecular weight (molar mass) of some common substances. The molar mass of a substance is the mass in grams of 1 mole of the substance. So, in the molecular form, there are 2 hydrogen atoms. The structure of molecular hydrogen is h−h . Hydrogen (h), 1.00794 g/mol ;
Hydrogen (h), 1.00794 g/mol ; Using the periodic table of the elements to find atomic weights, we find that hydrogen has an atomic weight of 1, and oxygen's is 16. The sum of the atomic masses of all atoms in a molecule, based on a scale in which the atomic masses of hydrogen, carbon, nitrogen, and oxygen are 1, 12, . The molecular weight of a hydrogen molecule is m=2.016×10−3 kg/mol. If we know the formula of a compound, determining the molar mass is simple.

Hydrogen, h · 2.016 ;
In order to calculate the . Hydrogen molar mass is 1.00794 g/mol · the molar mass of the hydrogen molecule is 2.016 g/mol. Carbon (c), 12.0107 g/mol ; Definition and molecular weight (molar mass) of some common substances. Using the periodic table of the elements to find atomic weights, we find that hydrogen has an atomic weight of 1, and oxygen's is 16. So, in the molecular form, there are 2 hydrogen atoms. C2h6, 2 atoms of carbon combine with 6 atoms of hydrogen to form ethane. The molecular weight of a hydrogen molecule is m=2.016×10−3 kg/mol. Iron (fe), 55.845 g/mol ; The sum of the atomic masses of all atoms in a molecule, based on a scale in which the atomic masses of hydrogen, carbon, nitrogen, and oxygen are 1, 12, . If we know the formula of a compound, determining the molar mass is simple. Rounded to three decimal places, the atomic weight of hydrogen is 1.008 amu and that of oxygen is 15.999 amu. Hydrogen (h), 1.00794 g/mol ;
Iron (fe), 55.845 g/mol ; Definition and molecular weight (molar mass) of some common substances. C2h6, 2 atoms of carbon combine with 6 atoms of hydrogen to form ethane. If we know the formula of a compound, determining the molar mass is simple. 1, h · hydrogen, 1.00797 ;

The structure of molecular hydrogen is h−h .
If we know the formula of a compound, determining the molar mass is simple. So, one more electron is needed to complete its octet. Using the periodic table of the elements to find atomic weights, we find that hydrogen has an atomic weight of 1, and oxygen's is 16. · hydrogen is a diatomic molecule with linear molecular geometry . Hydrogen molar mass is 1.00794 g/mol · the molar mass of the hydrogen molecule is 2.016 g/mol. Carbon (c), 12.0107 g/mol ; Rounded to three decimal places, the atomic weight of hydrogen is 1.008 amu and that of oxygen is 15.999 amu. The sum of the atomic masses of all atoms in a molecule, based on a scale in which the atomic masses of hydrogen, carbon, nitrogen, and oxygen are 1, 12, . 1, h · hydrogen, 1.00797 ; C2h6, 2 atoms of carbon combine with 6 atoms of hydrogen to form ethane. Hydrogen, h · 2.016 ; The structure of molecular hydrogen is h−h . In order to calculate the .
Molecular Mass Hydrogen - #302. Definition and molecular weight (molar mass) of some common substances. The molar mass of a substance is the mass in grams of 1 mole of the substance. If we know the formula of a compound, determining the molar mass is simple. Hydrogen, h · 2.016 ; Using the periodic table of the elements to find atomic weights, we find that hydrogen has an atomic weight of 1, and oxygen's is 16.
Komentar
Posting Komentar