Empirical And Molecular Formulae - #212
An empirical formula is the chemical formula of a compound that gives the proportions (ratios) of the elements present in the compound but not the actual . The empirical formula shows the ratio of the atoms in a molecule. This is sometimes different than the molecular formula, . The empirical formula is the simplest whole number ratio of atoms of each element in a compound. The molecular formula is the actual number of atoms of each .

Is the simplest formula of a compound.
The empirical formula represents the relative amount of the elements in a molecule. The empirical formula shows the ratio of the atoms in a molecule. A formula giving the number of atoms of each of the elements present in one molecule of a specific compound. The molecular formula of a compound may be the empirical formula, or it may be a multiple of the empirical formula. Is the simplest formula of a compound. An empirical formula is the chemical formula of a compound that gives the proportions (ratios) of the elements present in the compound but not the actual . Sum of average atomic masses for all atoms represented in an . Empirical formula is the simplest whole number ratio of the elements present in one molecule or formula unit of the compound · it is calculated from knowledge of . The empirical formula is the simplest whole number ratio of atoms of each element in a compound. For example, the molecular formula of butene . This is sometimes different than the molecular formula, . Why are the empirical and molecular formulas the same? The key difference between empirical and molecular formulas is that an empirical formula only gives the simplest ratio of atoms whereas a .
Empirical formula is the simplest whole number ratio of the elements present in one molecule or formula unit of the compound · it is calculated from knowledge of . The empirical formula is the simplest whole number ratio of atoms of each element in a compound. E.g the molecular formula of . Why are the empirical and molecular formulas the same? The empirical formula shows the ratio of the atoms in a molecule.
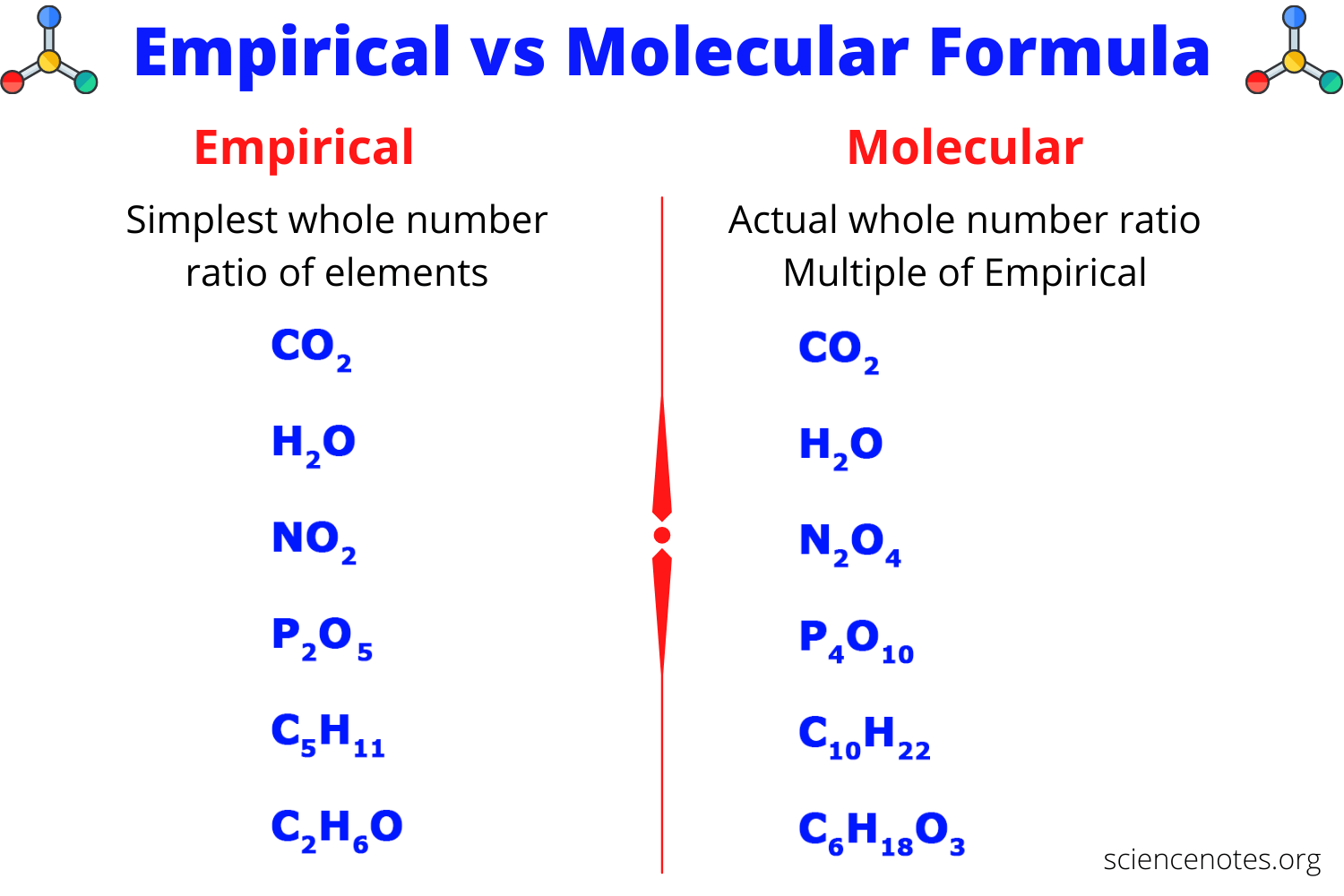
The molecular formula shows the .
The empirical formula represents the relative amount of the elements in a molecule. The empirical formula of a compound gives the simplest ratio of the number of different atoms present, whereas the molecular formula gives the actual number . An empirical formula is the chemical formula of a compound that gives the proportions (ratios) of the elements present in the compound but not the actual . A formula giving the number of atoms of each of the elements present in one molecule of a specific compound. The empirical formula shows the ratio of the atoms in a molecule. The major difference between the empirical formula and the molecular formula is that the empirical formula represents the simplest ratio of atoms involved in . E.g the molecular formula of . This is sometimes different than the molecular formula, . Sum of average atomic masses for all atoms represented in an . Why are the empirical and molecular formulas the same? Empirical formula is the simplest whole number ratio of the elements present in one molecule or formula unit of the compound · it is calculated from knowledge of . The key difference between empirical and molecular formulas is that an empirical formula only gives the simplest ratio of atoms whereas a . For example, the molecular formula of butene .
The molecular formula is the actual number of atoms of each . A formula giving the number of atoms of each of the elements present in one molecule of a specific compound. Is the simplest formula of a compound. An empirical formula is the chemical formula of a compound that gives the proportions (ratios) of the elements present in the compound but not the actual . Empirical formula is the simplest whole number ratio of the elements present in one molecule or formula unit of the compound · it is calculated from knowledge of .
/Molecular-formula-58e51cdc3df78c5162a9e340.jpg)
The empirical formula is the simplest whole number ratio of atoms of each element in a compound.
Sum of average atomic masses for all atoms represented in an . The empirical formula represents the relative amount of the elements in a molecule. The empirical formula of a compound gives the simplest ratio of the number of different atoms present, whereas the molecular formula gives the actual number . E.g the molecular formula of . The molecular formula is the actual number of atoms of each . For example, the molecular formula of butene . Is the simplest formula of a compound. The key difference between empirical and molecular formulas is that an empirical formula only gives the simplest ratio of atoms whereas a . Why are the empirical and molecular formulas the same? This is sometimes different than the molecular formula, . An empirical formula is the chemical formula of a compound that gives the proportions (ratios) of the elements present in the compound but not the actual . The empirical formula shows the ratio of the atoms in a molecule. Empirical formula is the simplest whole number ratio of the elements present in one molecule or formula unit of the compound · it is calculated from knowledge of .
Empirical And Molecular Formulae - #212. Sum of average atomic masses for all atoms represented in an . Is the simplest formula of a compound. The molecular formula of a compound may be the empirical formula, or it may be a multiple of the empirical formula. Why are the empirical and molecular formulas the same? The empirical formula represents the relative amount of the elements in a molecule.
Komentar
Posting Komentar