Balancing Molecular Equations - #185
Introduction to balancing equations and reactions. Anybody having chemistry as a subject is bound to come across balancing chemical equations. This chemistry video shows you how to balance chemical equations especially if you come across a fraction or an equation with polyatomic . If you are given a molecule of co2 it means that there is 1 carbon atom bonded to 2 oxygen atoms. A balanced equation is an equation for a chemical reaction in which the number of atoms for each element in the reaction and the total charge .

Sicl4 + h2o → h4sio4 + hcl · 3.
Conclusion · a balanced chemical equation has the same number of atoms of each type on both sides of the reaction, i.e., on the reactant as well as product side. Co2 + h2o → c6h12o6 + o · 2. Balanced equations are those whose coefficients result in equal numbers of atoms for each element in the reactants and products. Al + hcl → alcl3 + h · 4. This chemistry video shows you how to balance chemical equations especially if you come across a fraction or an equation with polyatomic . If you are given a molecule of co2 it means that there is 1 carbon atom bonded to 2 oxygen atoms. Introduction to balancing equations and reactions. Anybody having chemistry as a subject is bound to come across balancing chemical equations. In order to balance the chemical equation, you need to make sure the number of atoms of each element on the reactant side is equal to the number of atoms of . Thus, the mass of substances produced in a chemical reaction is always equal to the mass of reacting substances. Sicl4 + h2o → h4sio4 + hcl · 3. A balanced equation is an equation for a chemical reaction in which the number of atoms for each element in the reaction and the total charge . Chemical equation balancer · na + o2 = na2o in order for this equation to be balanced, there must be an equal amount of na on the left hand side as on the right .
Na2co3 + hcl → nacl + h2o + co . Thus, the mass of substances produced in a chemical reaction is always equal to the mass of reacting substances. Chemical equation balancer · na + o2 = na2o in order for this equation to be balanced, there must be an equal amount of na on the left hand side as on the right . Therefore, you need to have the . Al + hcl → alcl3 + h · 4.
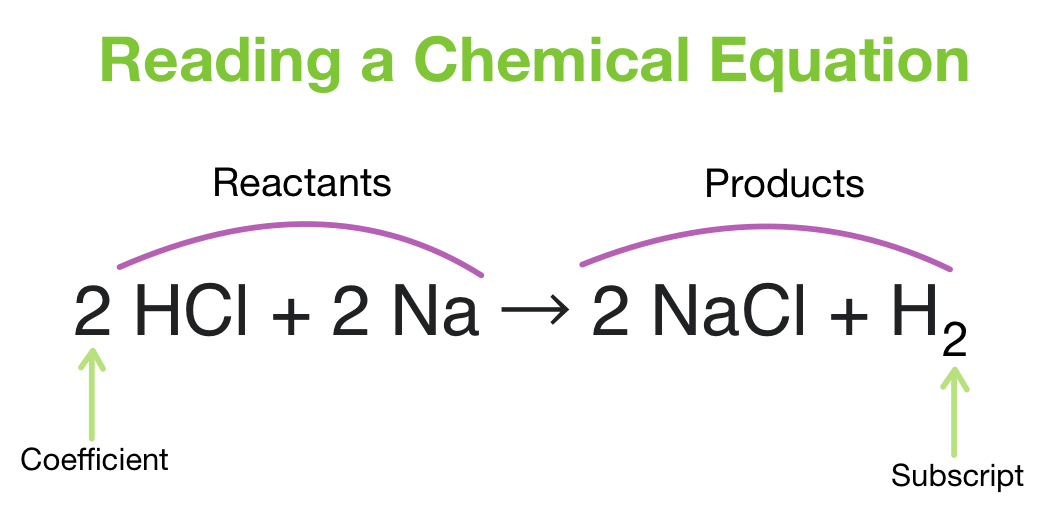
Co2 + h2o → c6h12o6 + o · 2.
Balanced equations are those whose coefficients result in equal numbers of atoms for each element in the reactants and products. This chemistry video shows you how to balance chemical equations especially if you come across a fraction or an equation with polyatomic . Sicl4 + h2o → h4sio4 + hcl · 3. In order to balance the chemical equation, you need to make sure the number of atoms of each element on the reactant side is equal to the number of atoms of . Co2 + h2o → c6h12o6 + o · 2. A balanced equation is an equation for a chemical reaction in which the number of atoms for each element in the reaction and the total charge . Therefore, you need to have the . Conclusion · a balanced chemical equation has the same number of atoms of each type on both sides of the reaction, i.e., on the reactant as well as product side. Na2co3 + hcl → nacl + h2o + co . Introduction to balancing equations and reactions. Anybody having chemistry as a subject is bound to come across balancing chemical equations. Thus, the mass of substances produced in a chemical reaction is always equal to the mass of reacting substances. If you are given a molecule of co2 it means that there is 1 carbon atom bonded to 2 oxygen atoms.
If you are given a molecule of co2 it means that there is 1 carbon atom bonded to 2 oxygen atoms. Anybody having chemistry as a subject is bound to come across balancing chemical equations. Thus, the mass of substances produced in a chemical reaction is always equal to the mass of reacting substances. This chemistry video shows you how to balance chemical equations especially if you come across a fraction or an equation with polyatomic . Na2co3 + hcl → nacl + h2o + co .

In order to balance the chemical equation, you need to make sure the number of atoms of each element on the reactant side is equal to the number of atoms of .
Sicl4 + h2o → h4sio4 + hcl · 3. Balanced equations are those whose coefficients result in equal numbers of atoms for each element in the reactants and products. In order to balance the chemical equation, you need to make sure the number of atoms of each element on the reactant side is equal to the number of atoms of . Therefore, you need to have the . Co2 + h2o → c6h12o6 + o · 2. A balanced equation is an equation for a chemical reaction in which the number of atoms for each element in the reaction and the total charge . If you are given a molecule of co2 it means that there is 1 carbon atom bonded to 2 oxygen atoms. Na2co3 + hcl → nacl + h2o + co . Anybody having chemistry as a subject is bound to come across balancing chemical equations. Al + hcl → alcl3 + h · 4. Introduction to balancing equations and reactions. Chemical equation balancer · na + o2 = na2o in order for this equation to be balanced, there must be an equal amount of na on the left hand side as on the right . Conclusion · a balanced chemical equation has the same number of atoms of each type on both sides of the reaction, i.e., on the reactant as well as product side.
Balancing Molecular Equations - #185. Sicl4 + h2o → h4sio4 + hcl · 3. Na2co3 + hcl → nacl + h2o + co . Al + hcl → alcl3 + h · 4. Conclusion · a balanced chemical equation has the same number of atoms of each type on both sides of the reaction, i.e., on the reactant as well as product side. If you are given a molecule of co2 it means that there is 1 carbon atom bonded to 2 oxygen atoms.
Komentar
Posting Komentar